
Chinese Journal OF Rice Science ›› 2023, Vol. 37 ›› Issue (4): 368-378.DOI: 10.16819/j.1001-7216.2023.221112
• Research Papers • Previous Articles Next Articles
WANG Shengyong1, CHEN Yuhang1, CHEN Huili1, HUANG Yujie1, ZHANG Xiaotian1, DING Shuangcheng2( ), WANG Hongwei1(
), WANG Hongwei1( )
)
Received:2022-11-24
Revised:2023-02-07
Online:2023-07-10
Published:2023-07-17
Contact:
*email: shchding@yangtzeu.edu.cn;wanghw@yangtzeu.edu.cn
汪胜勇1, 陈宇航1, 陈会丽1, 黄钰杰1, 张啸天1, 丁双成2( ), 王宏伟1(
), 王宏伟1( )
)
通讯作者:
*email: shchding@yangtzeu.edu.cn;wanghw@yangtzeu.edu.cn
基金资助:WANG Shengyong, CHEN Yuhang, CHEN Huili, HUANG Yujie, ZHANG Xiaotian, DING Shuangcheng, WANG Hongwei. Effects of High Temperature on Phenylpropane Metabolism and Downstream Branch Metabolic Pathways in Rice Meiosis[J]. Chinese Journal OF Rice Science, 2023, 37(4): 368-378.
汪胜勇, 陈宇航, 陈会丽, 黄钰杰, 张啸天, 丁双成, 王宏伟. 水稻减数分裂期高温对苯丙烷类代谢及下游分支代谢途径的影响[J]. 中国水稻科学, 2023, 37(4): 368-378.
Add to citation manager EndNote|Ris|BibTeX
URL: http://www.ricesci.cn/EN/10.16819/j.1001-7216.2023.221112
| 基因位点号(基因名) Locus ID(gene name) | 正向引物序列 Forward(5'-3') | 反向引物 Reverse(5'-3') |
|---|---|---|
| LOC_Os09g08072(INV1) | AAGAGCAGGGGTGTACAAG | AAGCTGTGAGTCTGTGGCTC |
| LOC_Os06g09450(SUS2) | GTGTGCTTGACACCATCCAC | CATGCGGAGACAGGATAACA |
| LOC_Os05g45420(SnRK1A) | ACAACCAGTGGCTACCTTGG | CGATGATCAGTGGCTGAGTT |
| LOC_Os07g09610(SnRK1B) | ATATCAGGCGCCGAATACTG | TGTGCCTGAAGAACTTGCTG |
| LOC_Os05g14550(TOR) | GCTGAACGCTGCAATGACTA | ACCGAACAAGTACTGGAGCA |
| NC_001320.1(ATPase) | TCGGTGGAGCTACTCTTGGA | CGGGCGCGGATCTATGAATA |
| Tubulin | TACCGTGCCCTTACTGTTCC | CGGTGGAATGTCACAGACAC |
Table 1. Primers used in gene expression analysis.
| 基因位点号(基因名) Locus ID(gene name) | 正向引物序列 Forward(5'-3') | 反向引物 Reverse(5'-3') |
|---|---|---|
| LOC_Os09g08072(INV1) | AAGAGCAGGGGTGTACAAG | AAGCTGTGAGTCTGTGGCTC |
| LOC_Os06g09450(SUS2) | GTGTGCTTGACACCATCCAC | CATGCGGAGACAGGATAACA |
| LOC_Os05g45420(SnRK1A) | ACAACCAGTGGCTACCTTGG | CGATGATCAGTGGCTGAGTT |
| LOC_Os07g09610(SnRK1B) | ATATCAGGCGCCGAATACTG | TGTGCCTGAAGAACTTGCTG |
| LOC_Os05g14550(TOR) | GCTGAACGCTGCAATGACTA | ACCGAACAAGTACTGGAGCA |
| NC_001320.1(ATPase) | TCGGTGGAGCTACTCTTGGA | CGGGCGCGGATCTATGAATA |
| Tubulin | TACCGTGCCCTTACTGTTCC | CGGTGGAATGTCACAGACAC |
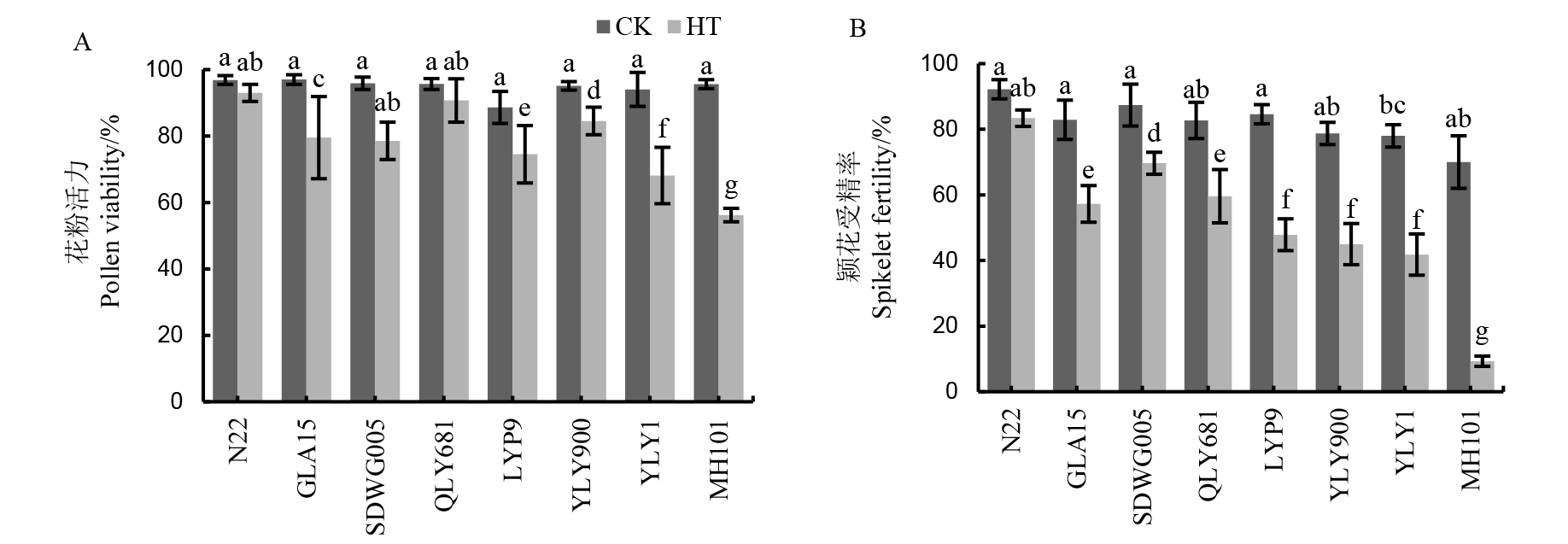
Fig. 2. Pollen vitality (A) and spikelet fertility (B) of different rice cultivars under HT during meiosis stage. GLA15, Guanglu’ai 15; QLY681, Quanliangyou 681; LYP9, Liangyoupeijiu; YLY900, Y Liangyou 900; YLY1, Y Liangyou 1; MH101, Mianhui 101. Mean ± standard deviation, n = 3; different lowercase letters on the bars indicate significant difference among varieties and treatments at 5% level. The same below.
| 相关系数 Correlation coefficient | 花粉活力 Pollen vitality | 颖花受精率 Spikelet fertility | 苯丙氨酸 解氨酶活性 Phenylalanine ammonia lyase activity | 肉桂酸-4- 羟化酶活性 Cinnamic acid- 4-hydroxylase activity | 4-香豆酸辅酶 A连接酶活性 4-Coumaric acid coenzyme A ligase activity | 总酚 Total phenols | 类黄酮 Flavonoids | 木质素 Lignin | |
|---|---|---|---|---|---|---|---|---|---|
| CK | 花粉活力Pollen viability | 1.000 | |||||||
| 颖花受精率Spikelet fertility | 0.615 | 1.000 | |||||||
| 苯丙氨酸解氨酶活性 Phenylalanine ammonialyase activity | 0.193 | 0.662 | 1.000 | ||||||
| 肉桂酸-4-羟化酶活性 Cinnamic acid-4-hydroxylase activity | 0.499 | 0.505 | 0.545 | 1.000 | |||||
| 4-香豆酸辅酶A连接酶活性 4-Coumaric acid coenzyme A ligase activity | 0.406 | 0.634 | 0.910** | 0.648 | 1.000 | ||||
| 总酚Total phenols | 0.446 | 0.596 | 0.836** | 0.840** | 0.930** | 1.000 | |||
| 总黄酮Flavonoids | 0.253 | 0.510 | 0.872** | 0.412 | 0.832** | 0.709* | 1.000 | ||
| 木质素Lignin | 0.334 | 0.576 | 0.918** | 0.632 | 0.904** | 0.864** | 0.960** | 1.000 | |
| HT | 花粉活力Pollen vitality | 1.000 | |||||||
| 颖花受精率Spikelet fertility | 0.905** | 1.000 | |||||||
| 苯丙氨酸解氨酶活性 Phenylalanine ammonialyase activity | 0.188 | 0.499 | 1.000 | ||||||
| 肉桂酸-4-羟化酶活性 Cinnamic acid-4-hydroxylase activity | 0.838** | 0.694 | 0.068 | 1.000 | |||||
| 4-香豆酸辅酶A连接酶活性 4-Coumaric acid coenzyme A ligase activity | 0.416 | 0.403 | 0.485 | 0.546 | 1.000 | ||||
| 总酚Total phenols | 0.561 | 0.629 | 0.539 | 0.539 | 0.693 | 1.000 | |||
| 总黄酮Flavonoids | 0.562 | 0.588 | 0.659 | 0.406 | 0.733* | 0.662 | 1.000 | ||
| 木质素Lignin | 0.775* | 0.887** | 0.649 | 0.598 | 0.590 | 0.668 | 0.860** | 1.000 | |
Table 2. Correlation analysis of various indexes involved in phenylpropane metabolism and downstream branch metabolic pathways under CK and HT.
| 相关系数 Correlation coefficient | 花粉活力 Pollen vitality | 颖花受精率 Spikelet fertility | 苯丙氨酸 解氨酶活性 Phenylalanine ammonia lyase activity | 肉桂酸-4- 羟化酶活性 Cinnamic acid- 4-hydroxylase activity | 4-香豆酸辅酶 A连接酶活性 4-Coumaric acid coenzyme A ligase activity | 总酚 Total phenols | 类黄酮 Flavonoids | 木质素 Lignin | |
|---|---|---|---|---|---|---|---|---|---|
| CK | 花粉活力Pollen viability | 1.000 | |||||||
| 颖花受精率Spikelet fertility | 0.615 | 1.000 | |||||||
| 苯丙氨酸解氨酶活性 Phenylalanine ammonialyase activity | 0.193 | 0.662 | 1.000 | ||||||
| 肉桂酸-4-羟化酶活性 Cinnamic acid-4-hydroxylase activity | 0.499 | 0.505 | 0.545 | 1.000 | |||||
| 4-香豆酸辅酶A连接酶活性 4-Coumaric acid coenzyme A ligase activity | 0.406 | 0.634 | 0.910** | 0.648 | 1.000 | ||||
| 总酚Total phenols | 0.446 | 0.596 | 0.836** | 0.840** | 0.930** | 1.000 | |||
| 总黄酮Flavonoids | 0.253 | 0.510 | 0.872** | 0.412 | 0.832** | 0.709* | 1.000 | ||
| 木质素Lignin | 0.334 | 0.576 | 0.918** | 0.632 | 0.904** | 0.864** | 0.960** | 1.000 | |
| HT | 花粉活力Pollen vitality | 1.000 | |||||||
| 颖花受精率Spikelet fertility | 0.905** | 1.000 | |||||||
| 苯丙氨酸解氨酶活性 Phenylalanine ammonialyase activity | 0.188 | 0.499 | 1.000 | ||||||
| 肉桂酸-4-羟化酶活性 Cinnamic acid-4-hydroxylase activity | 0.838** | 0.694 | 0.068 | 1.000 | |||||
| 4-香豆酸辅酶A连接酶活性 4-Coumaric acid coenzyme A ligase activity | 0.416 | 0.403 | 0.485 | 0.546 | 1.000 | ||||
| 总酚Total phenols | 0.561 | 0.629 | 0.539 | 0.539 | 0.693 | 1.000 | |||
| 总黄酮Flavonoids | 0.562 | 0.588 | 0.659 | 0.406 | 0.733* | 0.662 | 1.000 | ||
| 木质素Lignin | 0.775* | 0.887** | 0.649 | 0.598 | 0.590 | 0.668 | 0.860** | 1.000 | |
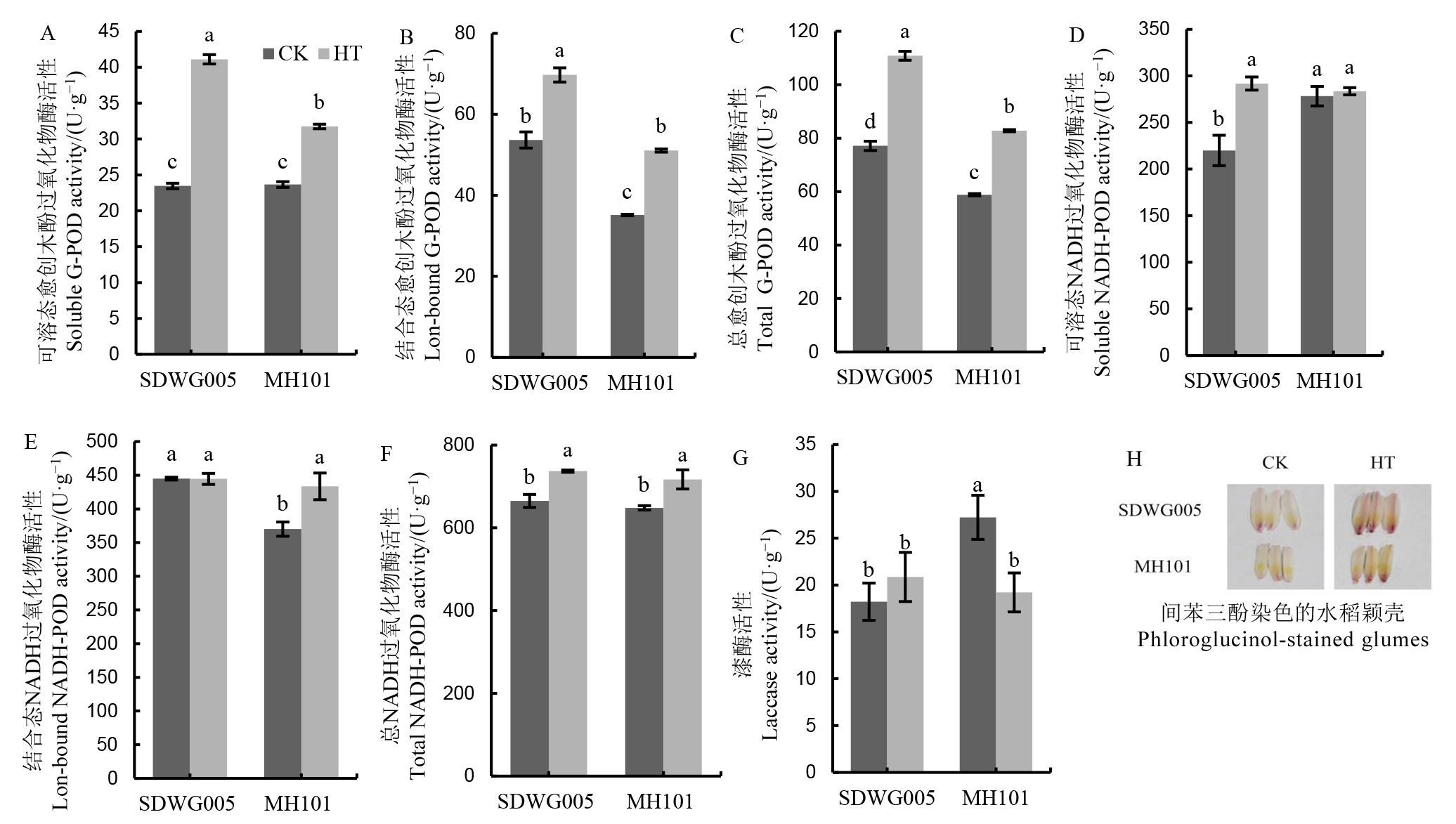
Fig. 5. Effects of HT on cell wall peoxidase activities in spikelets during meiosis stage. Different lowercase letters above the bars indicate significant difference among different varieties and treatments at 5% level.
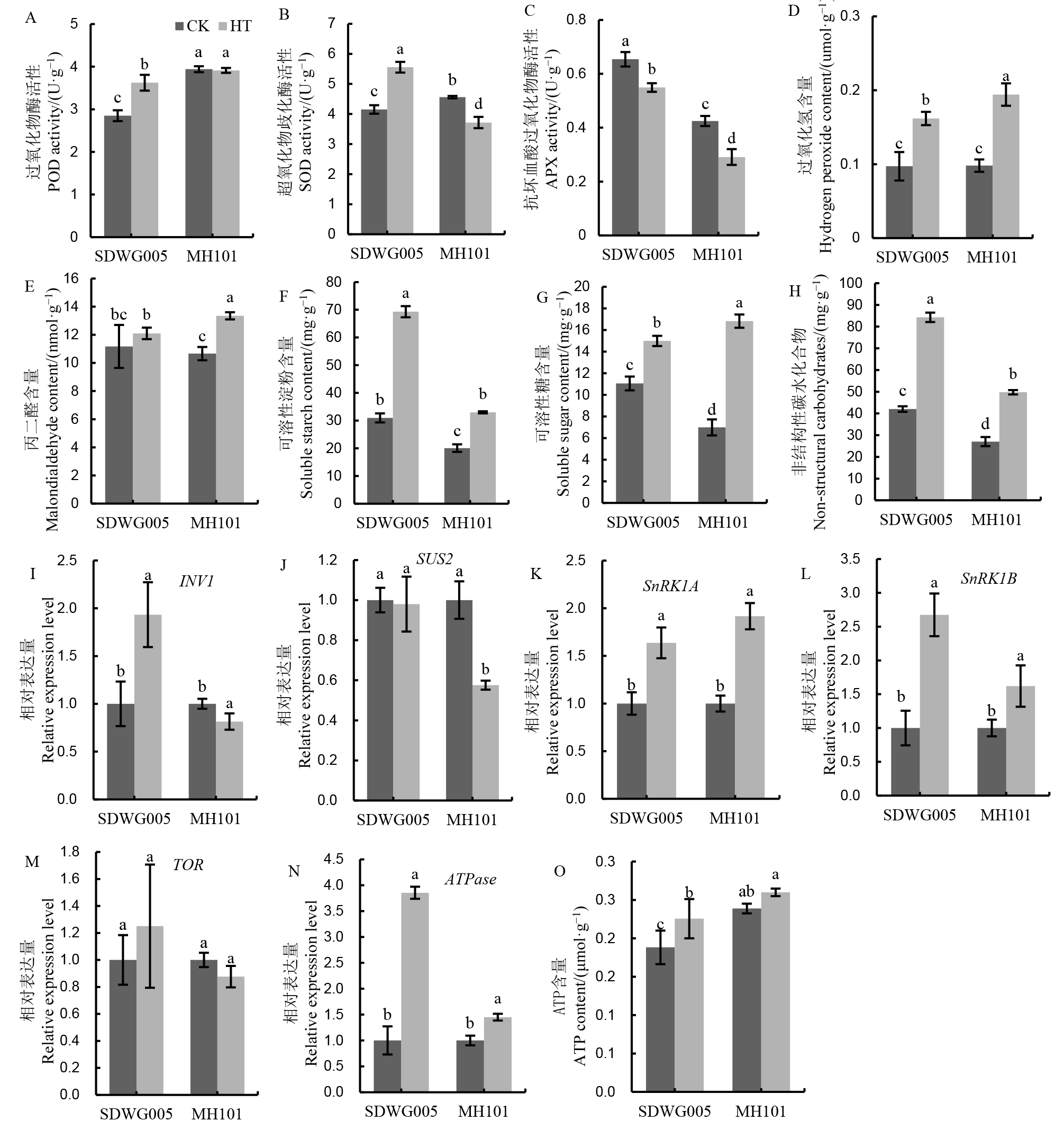
Fig. 6. Effects of HT on antioxidant enzyme activities, H2O2, malondialdehyde, carbohydrate contents, and gene expression involved in sucrose metabolic and energy level in spikelets during meiosis stage. Different lowercase letters above the bars indicate significant difference among different varieties and treatments at 5% level.
| [1] | Shi P H, Tang L, Wang L H, Sun T, Liu L L, Cao W X, Zhu Y. Post-heading heat stress in rice of south China during 1981-2010[J]. PLoS One, 2015, 10(6): e0130642. |
| [2] | Shukla P R, Skeg J, Calvo Buendia E, Masson-Delmotte V, Pörtner H O, Malley J. Climate change and land:An IPCC special report on climate change, desertification, land degradation, sustainable land management, food security, and greenhouse gas fluxes in terrestrial ecosystems[M]. Koyoto, Japan: Intergovernmental Panel on Climate Change(IPCC), 2019. |
| [3] | 杨军, 章毅之, 贺浩华, 李迎春, 陈小荣, 边建民, 金国花, 李翔翔, 黄淑娥. 水稻高温热害的研究现状与进展[J]. 应用生态学报, 2020, 31(8): 2817-2830. |
| Yang J, Zhang Y Z, He H H, Li Y C, Chen X R, Bian J M, Jin G H, Li X X, Huang S E. Current status and research advances of high-temperature hazards in rice[J]. Chinese Journal of Applied Ecology, 2020, 31(8): 2817-2830. (in Chinese with English abstract) | |
| [4] | 田小海, 罗海伟, 吴晨阳. 中国水稻热害研究历史、进展与展望[J]. 中国农学通报, 2009, 25(22): 166-168. |
| Tian X H, Luo H W, Wu C Y. Research on heat stress of rice in China: Progress and prospect[J]. Chinese Agricultural Science Bulletin, 2009, 25(22): 166-168. (in Chinese with English abstract) | |
| [5] | Peng S B, Huang J L, Sheehy J E, Laza R C, Cassman K G. Rice yields decline with higher night temperature from global warming[J]. Proceedings of the National Academy of Sciences of the United States of America, 2004, 101(27): 9971-9975. |
| [6] | Ray D K, Gerber J S, Macdonald G K, West P C. Climate variation explains a third of global crop yield variability[J]. Nature Communications, 2015, 6: 5989. |
| [7] | Sage T L, Bagha S, Lundsgaard-Nielsen V, Branch H A, Sultmanis S, Sage R F. The effect of high temperature stress on male and female reproduction in plants[J]. Field Crops Research, 2015, 182: 30-42. |
| [8] | Endo M, Tsuchiya T, Hamada K, Kawamura S, Yano K. High temperatures cause male sterility in rice plants with transcriptional alterations during pollen development[J]. Plant & Cell Physiology, 2009, 50(11): 1911-22. |
| [9] | Sato S, Peet M M, Thomas J F. Determining critical pre- and post-anthesis periods and physiological processes in Lycopersicon esculentum Mill. exposed to moderately elevated temperatures[J]. Journal of Experimental Botany, 2002, 53(371): 1187-1195. |
| [10] | Jagadish S V K, Craufurd P Q, Wheeler T R. Phenotyping parents of mapping populations of rice for heat tolerance during anthesis[J]. Crop Science, 2008, 48(3): 1140-1146. |
| [11] | Wilson Z A, Song J, Taylor B, Yang C Y. The final split: The regulation of anther dehiscence[J]. Journal of Experimental Botany, 2011, 62(5): 1633-1649. |
| [12] | Matsui T, Omasa K, Horie T. High temperature at flowering inhibits swelling of pollen grains, a driving force for thecae dehiscence in rice (Oryza sativa L.)[J]. Plant Production Science, 2000, 3(4): 430-434. |
| [13] | Matsui T, Omasa K. Rice cultivars tolerant to high temperature at flowering: Anther characteristics[J]. Annals of Botany, 2002, 89(6): 683-687. |
| [14] | Prasad P V V, Boote K J, Allen L H, Sheehy J E. Species, ecotype and cultivar differences in spikelet fertility and harvest index of rice in response to high temperature[J]. Field Crops Research, 2006, 95(2): 398-411. |
| [15] | Jagadish S V, Muthurajan R, Oane R, Wheeler T R, Heuer S, Bennett J, Craufurd P Q. Physiological and proteomic approaches to address heat tolerance during anthesis in rice (Oryza sativa L.)[J]. Journal of Experimental Botany, 2010, 61(1): 143-156. |
| [16] | Kobayashi K, Matsui T, Murata Y, Yamamoto M. Percentage of dehisced thecae and length of dehiscence control pollination stability of rice cultivars[J]. Plant Production Science, 2011, 14(2): 89-95. |
| [17] | 张桂莲, 张顺堂, 肖浪涛, 唐文帮, 肖应辉, 陈立云. 抽穗开花期高温胁迫对水稻花药、花粉粒及柱头生理特性的影响[J]. 中国水稻科学, 2014, 28(2): 155-166. |
| Zhang G L, Zhang S T, Xiao L T, Tang W B, Xiao Y H, Chen L Y. Effect of high temperature stress on physiological characteristics of anther, pollen and stigma of rice during heading-flowering stage[J]. Chinese Journal of Rice Science, 2014, 28(2): 155-166. (in Chinese with English abstract) | |
| [18] | Wahid A, Gelani S, Ashraf M, Foolad M R. Heat tolerance in plants: An overview[J]. Environmental and Experimental Botany, 2007, 61(3): 199-223. |
| [19] | Locato V, de Pinto M C, de Gara L. Different involvement of the mitochondrial, plastidial and cytosolic ascorbate-glutathione redox enzymes in heat shock responses[J]. Physiologia Plantarum, 2009, 135(3): 296-306. |
| [20] | Locato V, Gadaleta C, de Gara L, de Pinto M C. Production of reactive species and modulation of antioxidant network in response to heat shock: A critical balance for cell fate[J]. Plant Cell & Environment, 2008, 31(11): 1606-1619. |
| [21] | Parish R W, Phan H A, Iacuone S, Li S F. Tapetal development and abiotic stress[J]. Functional Plant Biology, 2012, 39(7): 553-559. |
| [22] | De Storme N, Geelen D. The impact of environmental stress on male reproductive development in plants: Biological processes and molecular mechanisms[J]. Plant Cell & Environment, 2014, 37(1): 1-18. |
| [23] | Bahuguna R N, Solis C A, Shi W J, Jagadish K S. Post-flowering night respiration and altered sink activity account for high night temperature-induced grain yield and quality loss in rice (Oryza sativa L.)[J]. Physiologia Plantarum, 2017, 159(1): 59-73. |
| [24] | Herrmann K M, Weaver L M. The Shikimate Pathway[J]. Annual Review of Plant Physiology and Plant Molecular Biology, 1999, 50: 473-503. |
| [25] | Schubert W J, Acerbo S N. The conversion of D-glucose into lignin in Norwegian spruce[J]. Archives of Biochemistry and Biophysics, 1959, 83(1): 178-82. |
| [26] | Rogers L A, Dubos C, Cullis I F, Surman C, Poole M, Willment J, Mansfield S D, Campbell M M. Light, the circadian clock, and sugar perception in the control of lignin biosynthesis[J]. Journal of Experimental Botany, 2005, 56(416): 1651-63. |
| [27] | Jiao Y, Gong X, Qi K, Xie Z, Wang Y, Yuan K, Pan Q, Zhang S, Shiratake K, Khanizadeh S, Tao S. Transcriptome analysis provides new ideas for studying the regulation of glucose-induced lignin biosynthesis in pear calli[J]. BMC Plant Biology, 2022, 22(1): 310. |
| [28] | Poovaiah C R, Mazarei M, Decker S R, Turner G B, Sykes R W, Davis M F, Stewart C N, Jr. Transgenic switchgrass biomass is increased by overexpression of switchgrass sucrose synthase (PvSUS1)[J]. Biotechnology Journal, 2015, 10(4): 552-563. |
| [29] | Dong N Q, Lin H X. Contribution of phenylpropanoid metabolism to plant development and plant-environment interactions[J]. Journal of Integrative Plant Biology, 2021, 63(1): 180-209. |
| [30] | Muhlemann J K, Younts T L B, Muday G K. Flavonols control pollen tube growth and integrity by regulating ROS homeostasis during high-temperature stress[J]. Proceedings of the National Academy of Sciences of the United States of America, 2018, 115(47): e11188-e11197. |
| [31] | Zhang D B, Wilson Z A. Stamen specification and anther development in rice[J]. Chinese Science Bulletin, 2009, 54(14): 2342-2353. |
| [32] | Fecht-Christoffers M M, Fuhrs H, Braun H P, Horst W J. The role of hydrogen peroxide-producing and hydrogen peroxide-consuming peroxidases in the leaf apoplast of cowpea in manganese tolerance[J]. Plant Physiology, 2006, 140(4): 1451-63. |
| [33] | Dong N Q, Sun Y, Guo T, Shi C L, Zhang Y M, Lin H X. UDP-glucosyltransferase regulates grain size and abiotic stress tolerance associated with metabolic flux redirection in rice[J]. Nature Communications, 2020, 11(1): 2629. |
| [34] | Liu Y Y, Ge Y H, Bi Y, Li C Y, Deng H W, Hu L G, Dong B. Effect of postharvest acibenzolar-S-methyl dipping on phenylpropanoid pathway metabolism in muskmelon fruits[J]. Scientia Horticulturae, 2014, 168: 113-119. |
| [35] | 张水明, 龚凌燕, 曹丹琴, 张永娟, 杨健. 石榴种皮总木质素含量及PgCOMT基因的克隆与表达[J]. 热带亚热带植物学报, 2015, 23(1): 65-73. |
| Zhang S M, Gong L Y, Cao D Q, Zhang Y J, Yang J. Total lignin content in pomegranate seed coat and cloning andexpression analysis of PgCOMT gene[J]. Journal of Tropical and Subtropical Botany, 2015, 23(1): 65-73. (in Chinese with English abstract) | |
| [36] | 黄晓彤, 史锐, 刘苗苗, 丛龙娇, 刘斯文, 王琪瑶. 同属不同种桑叶总黄酮的含量测定及分析[J]. 亚太传统医药, 2022, 18(8): 91-95. |
| Huang X T, Shi R, Liu M M, Cong L J, Liu S W, Wang Q Y. Optimization of extraction process and content determination of total flavonoids from different mulberry leaves[J]. Asia-Pacific Traditional Medicine, 2022, 18(8): 91-95. (in Chinese with English abstract) | |
| [37] | 王成章, 李建华, 郭玉霞, 方丽云, 高永革. 光周期对不同秋眠型苜蓿SOD、POD活性的影响[J]. 草地学报, 2007, 15(5): 407-411. (in Chinese with English abstract) |
| Wang C Z, Li J H, Guo Y X, Fang L Y, Gao Y G. Effect of photoperiod on SOD and POD activities in alfalfa varieties with different fall dormancy[J]. Acta Agrestia Sinica, 2007, 15(5): 407-411. | |
| [38] | Nakano Y, Asada K. Hydrogen peroxide is scavenged by ascorbate-specificperoxidase in spinach chloroplasts[J]. Plant and Cell Physiology, 1981, 22(5): 867-880. |
| [39] | 李光彦. 能量代谢影响水稻耐热性的作用机理[D]. 武汉: 华中农业大学, 2021. |
| Li G Y. The mechanism of energy metabolism mediating rice heat resistance[D]. Wuhan: Huangzhong Agricultural University, 2021. (in Chinese with English abstract) | |
| [40] | 刘清泉. 铜胁迫下水稻木质素合成的响应机制及水稻漆酶在植物重金属耐性中的作用[D]. 南京: 南京农业大学, 2015. |
| Liu Q Q. Response mechanism of lignin synthesis in rice under copper stress and the role of rice in plants tolerance to heavy metal[D]. Nanjing: Nanjing Agricultural University, 2015. (in Chinese with English abstract) | |
| [41] | 崔莹莹, 王晓玲. 水稻产量相关性状QTL的遗传研究进展[J]. 江苏农业科学, 2017, 45(13): 1-7. |
| Cui Y Y, Wang X L. Genetic research progress of QTL related traits in rice yield[J]. Jiangsu Agricultural Sciences, 2017, 45(13): 1-7. (in Chinese) | |
| [42] | 张桂莲, 张顺堂, 肖浪涛, 武小金, 肖应辉, 陈立云. 花期高温胁迫对水稻花药生理特性及花粉性状的影响[J]. 作物学报, 2013, 39(1): 177-183. |
| Zhang G L, Zhang S T, Xiao L T, Wu X J, Xiao Y H, Chen L Y. Effect of high temperature stress on physiological characteristics of anther and pollen traits of rice at flowering stage[J]. Acta Agronomica Sinica, 2013, 39(1): 177-183. (in Chinese with English abstract) | |
| [43] | Oshino T, Abiko M, Saito R, Higashitani A. Premature progression of anther early developmental programs accompanied by comprehensive alterations in transcription during high-temperature in barley plants[J]. Molecular Genetics and Genomics, 2007, 278(1): 31-42. |
| [44] | Feng B H, Zhang C X, Chen T T, Zhang X F, Tao L X, Fu G F. Salicylic acid reverses pollen abortion of rice caused by heat stress[J]. BMC Plant Biology, 2018, 18(1): 245. |
| [45] | Fraser C M, Chapple C. The phenylpropanoid pathway in Arabidopsis[J]. Arabidopsis Book, 2011, 9: e0152. |
| [46] | Dixon R A. Natural products and plant disease resistance[J]. Nature, 2001, 411(6839): 843-847. |
| [47] | Mishra A K, Baek K H. Salicylic acid biosynthesis and metabolism: A divergent pathway for plants and bacteria[J]. Biomolecules, 2021. 11(5): 705. |
| [48] | 陈爱国, 彭东, 陈向东, 石屹, 梁洪波. 烤烟苯丙烷代谢中相关酶活性和多酚产物的关系研究[C]. 山东植物生理学会第七次代表大会暨植物生物学与现代农业研讨会, 济南: 山东省科学技术协会, 2012: 240-246. |
| Chen A G, Peng D, Chen X D, Shi Y, Liang H B. Studies on Relationships of Related Enzyme Activities and Polyphenol Products of Phenylpropanoid Metabolic Pathway in Flue-cured tobacco[C]. Proceedings of the 7th Congress of Shandong Plant Physiology Society and Symposium on Plant Biology and Modern Agriculture, Jinan: Shandong Science and technology Association, 2012: 240-246. (in Chinese with English abstract) | |
| [49] | Zhang C X, Feng B H, Chen T T, Fu W M, Li H B, Li G Y, Jin Q Y, Fu G F. Heat stress-reduced kernel weight in rice at anthesis is associated with impaired source-sink relationship and sugars allocation[J]. Environmental and Experimental Botany, 2018, 155: 718-733. |
| [50] | Baena-González E, Hanson J. Shaping plant development through the SnRK1-TOR metabolic regulators[J]. Current Opinion in Plant Biology, 2017, 35: 152-157. |
| [51] | Yu P H, Jiang N, Fu W M, Zheng G J, Li G Y, Feng B H, Chen T T, Ma J Y, Li H B, Tao L X, Fu G F. ATP hydrolysis determines cold tolerance by regulating available energy for glutathione synthesis in rice seedling plants[J]. Rice, 2020, 13(1): 23. |
| [52] | Li G Y, Zhang C X, Zhang G H, Fu G F. Abscisic acid negatively modulates heat tolerance in rolled leaf rice by increasing leaf temperature and regulating energy homeostasis[J]. Rice, 2020, 13(1): 18. |
| [1] | GUO Zhan, ZHANG Yunbo. Research Progress in Physiological,Biochemical Responses of Rice to Drought Stress and Its Molecular Regulation [J]. Chinese Journal OF Rice Science, 2024, 38(4): 335-349. |
| [2] | WEI Huanhe, MA Weiyi, ZUO Boyuan, WANG Lulu, ZHU Wang, GENG Xiaoyu, ZHANG Xiang, MENG Tianyao, CHEN Yinglong, GAO Pinglei, XU Ke, HUO Zhongyang, DAI Qigen. Research Progress in the Effect of Salinity, Drought, and Their Combined Stresses on Rice Yield and Quality Formation [J]. Chinese Journal OF Rice Science, 2024, 38(4): 350-363. |
| [3] | XU Danjie, LIN Qiaoxia, LI Zhengkang, ZHUANG Xiaoqian, LING Yu, LAI Meiling, CHEN Xiaoting, LU Guodong. OsOPR10 Positively Regulates Rice Blast and Bacterial Blight Resistance [J]. Chinese Journal OF Rice Science, 2024, 38(4): 364-374. |
| [4] | CHEN Mingliang, ZENG Xihua, SHEN Yumin, LUO Shiyou, HU Lanxiang, XIONG Wentao, XIONG Huanjin, WU Xiaoyan, XIAO Yeqing. Typing of Inter-subspecific Fertility Loci and Fertility Locus Pattern of indica-japonica Hybrid Rice [J]. Chinese Journal OF Rice Science, 2024, 38(4): 386-396. |
| [5] | DING Zhengquan, PAN Yueyun, SHI Yang, HUANG Haixiang. Comprehensive Evaluation and Comparative Analysis of Jiahe Series Long-Grain japonica Rice with High Eating Quality Based on Gene Chip Technology [J]. Chinese Journal OF Rice Science, 2024, 38(4): 397-408. |
| [6] | HOU Xiaoqin, WANG Ying, YU Bei, FU Weimeng, FENG Baohua, SHEN Yichao, XIE Hangjun, WANG Huanran, XU Yongqiang, WU Zhihai, WANG Jianjun, TAO Longxing, FU Guanfu. Mechanisms Behind the Role of Potassium Fulvic Acid in Enhancing Salt Tolerance in Rice Seedlings [J]. Chinese Journal OF Rice Science, 2024, 38(4): 409-421. |
| [7] | LÜ Zhou, YI Binghuai, CHEN Pingping, ZHOU Wenxin, TANG Wenbang, YI Zhenxie. Effects of Nitrogen Application Rate and Transplanting Density on Yield Formation of Small Seed Hybrid Rice [J]. Chinese Journal OF Rice Science, 2024, 38(4): 422-436. |
| [8] | HU Jijie, HU Zhihua, ZHANG Junhua, CAO Xiaochuang, JIN Qianyu, ZHANG Zhiyuan, ZHU Lianfeng. Effects of Rhizosphere Saturated Dissolved Oxygen on Photosynthetic and Growth Characteristics of Rice at Tillering Stage [J]. Chinese Journal OF Rice Science, 2024, 38(4): 437-446. |
| [9] | WU Yue, LIANG Chengwei, ZHAO Chenfei, SUN Jian, MA Dianrong. Occurrence of Weedy Rice Disaster and Ecotype Evolution in Direct-Seeded Rice Fields [J]. Chinese Journal OF Rice Science, 2024, 38(4): 447-455. |
| [10] | LIU Fuxiang, ZHEN Haoyang, PENG Huan, ZHENG Liuchun, PENG Deliang, WEN Yanhua. Investigation and Species Identification of Cyst Nematode Disease on Rice in Guangdong Province [J]. Chinese Journal OF Rice Science, 2024, 38(4): 456-461. |
| [11] | CHEN Haotian, QIN Yuan, ZHONG Xiaohan, LIN Chenyu, QIN Jinghang, YANG Jianchang, ZHANG Weiyang. Research Progress on the Relationship Between Rice Root, Soil Properties and Methane Emissions in Paddy Fields [J]. Chinese Journal OF Rice Science, 2024, 38(3): 233-245. |
| [12] | MIAO Jun, RAN Jinhui, XU Mengbin, BO Liubing, WANG Ping, LIANG Guohua, ZHOU Yong. Overexpression of RGG2, a Heterotrimeric G Protein γ Subunit-Encoding Gene, Improves Drought Tolerance in Rice [J]. Chinese Journal OF Rice Science, 2024, 38(3): 246-255. |
| [13] | YIN Xiaoxiao, ZHANG Zhihan, YAN Xiulian, LIAO Rong, YANG Sijia, Beenish HASSAN, GUO Daiming, FAN Jing, ZHAO Zhixue, WANG Wenming. Signal Peptide Validation and Expression Analysis of Multiple Effectors from Ustilaginoidea virens [J]. Chinese Journal OF Rice Science, 2024, 38(3): 256-265. |
| [14] | ZHU Yujing, GUI Jinxin, GONG Chengyun, LUO Xinyang, SHI Jubin, ZHANG Haiqing, HE Jiwai. QTL Mapping for Tiller Angle in Rice by Genome-wide Association Analysis [J]. Chinese Journal OF Rice Science, 2024, 38(3): 266-276. |
| [15] | ZHAO Yiting, XIE Keran, GAO Ti, CUI Kehui. Effects of Drought Priming During Tillering Stage on Panicle Development and Yield Formation Under High Temperature During Panicle Initiation Stage in Rice [J]. Chinese Journal OF Rice Science, 2024, 38(3): 277-289. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||